ESCRT machinery
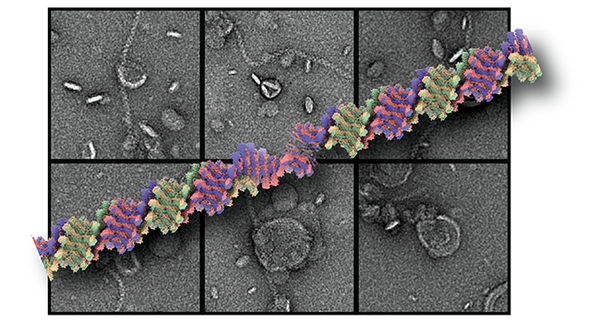
ESCRT machinery proteins mediate a range of cellular membrane remodeling activities that were initially described in multivesicular biogenesis of eukaryotes and henceforth received their designation endosomal sorting complexes for transport (ESCRT). In the meantime, we and others could show that ESCRT-III proteins have ancestors in bacteria and archaea (Junglas et al., 2021, Melnikov et al., 2025). Common all of these membrane-remodeling processes is the assembly of ESCRT-III subunits into polymeric structures. We elucidated the structural mechanism of assembly and membrane remodeling for cyanobacterial ESCRT-III proteins PspA and Vipp1 involved in bacterial membrane repair and thylakoid membrane biogenesis (Junglas et al., 2025, Junglas et al., 2025, Hudina et al., 2025). In eukaryotes, there are multiple isoforms that are known to form hetero-complex structures performing the central membrane remodeling steps. We determined the cryo-EM structure of a helical assembly of S. cerevisiae Vps24 at 3.2 Å resolution and found that Vps24 adopts an elongated open conformation (Huber et al., 2020). We study the biochemical properties and the structural biology of the ESCRT-III family proteins across the kingdom of life.
Related Publications
- Hudina, E., Schott-Verdugo, S., Junglas, B., Kutzner, M., Ritter, I., Hellmann, N., Schneider, D., Gohlke, H., Sachse, C., 2025. The bacterial ESCRT-III PspA rods thin lipid tubules and increase membrane curvature through helix α0 interactions. Proc Natl Acad Sci U S A 122, e2506286122.
- Junglas, B., Hudina, E., Schönnenbeck, P., Ritter, I., Heddier, A., Santiago-Schübel, B., Huesgen, P.F., Schneider, D., Sachse, C., 2025a. Structural plasticity of bacterial ESCRT-III protein PspA in higher-order assemblies. Nat Struct Mol Biol 32, 23–34.
- Junglas, B., Kartte, D., Kutzner, M., Hellmann, N., Ritter, I., Schneider, D., Sachse, C., 2025b. Structural basis for Vipp1 membrane binding: from loose coats and carpets to ring and rod assemblies. Nat Struct Mol Biol 32, 555–570.
- Melnikov, N., Junglas, B., Halbi, G., Nachmias, D., Zerbib, E., Gueta, N., Upcher, A., Zalk, R., Sachse, C., Bernheim-Groswasser, A., Elia, N., 2025. The Asgard archaeal ESCRT-III system forms helical filaments and remodels eukaryotic-like membranes. EMBO J 44, 665–681.
- Junglas B., Huber S.T., Heidler T., Schlösser R., Mann D., Hennig R., Clarke M., Hellmann N., Schneider D. and Sachse C. PspA adopts an ESCRT-III-like fold and remodels bacterial membranes. (2021) Cell 184(14): 3674–3688.e18
- Huber S.T., Mostafavi S., Mortensen S.A., Sachse C. Structure and assembly of ESCRT-III helical Vps24 filaments. (2020) Sci Advances 6(34): eaba4897.
