SynMech
A synaptic mechanogenetic technology to repair brain connectivity
Duration
February 2023 to January 2028
Contact

Dr.rer.nat Ali Mohammad Pourshahidi
PostDoctoral Researcher
Building 03.6 / Room 201
+49 2461/61-2691
E-Mail
Prof. Dr. Hans-Joachim Krause
PI, Group leader
Building 03.6 / Room 408
+49 2461/61-2955
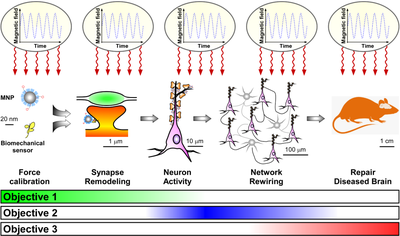
E-MailWe aim to develop a mechanogenetic technology to regulate functional connectivity of neural circuits, and show how it can be harnessed for therapeutic purposes in high-prevalence treatment-resistant brain disorders.
Mechanogenetics is an emerging field of health science that attempts to regulate neural networks by combining the advantages of optogenetics with those of magneto-mechanical stimulations; like optogenetics, it relies on targeted actuators to achieve circuit specificity, while exploiting magnetic fields to remotely stimulate the brain. Yet, despite solid theoretical foundations and encouraging experimental results, we are to date unable to repair a dysfunctional brain using mechanogenetics due to technological barriers in spatial resolution and in vivo implementation.
We propose an innovative solution based on functionalized biocompatible magnetic nanoparticles and bioengineered synaptic mechanosensors that synergistically integrate at specific synaptic connections to bidirectionally regulate brain circuit connectivity in response to focused magnetic fields of different frequencies delivered via high-permeability transcranial magnetic stimulators. By hijacking the signaling pathways of synaptic mechanosensors, we aim to promote a normalization of neural circuit activity that outlasts the therapeutic intervention. We will assess the capacity of the synaptic mechanogenetic toolkit to promote and depress network activity in mouse models of stroke and epilepsy.
To achieve this ambitious goal, we have gathered an interdisciplinary consortium going from material scientists and electronic experts to physiologists and clinicians. Our approach, based on magnetic fields that penetrate brain tissue unimpeded, is predicted to go beyond current therapeutic paradigms because it does not require implantation of invasive devices, and at the same time, promises to achieve subcellular resolution for repairing connectivity defects that underlie most brain disorders.


