In order to establish a “communication” with neuronal networks one must be able to "listen" first. Neurons are electrogenic cells, which produce electronic potentials, so-called action potentials (APs). These APs are the building blocks of neuronal communication and thus brain activity. Being able to record and stimulate the APs paves the way for understanding the brain on a deeper level. Modern micro/nano-electronics provide us with a set of devices to fulfil this purpose, starting with patch-clamp and microelectrode arrays (MEAs) and moving towards transistors and miniaturization.
Patch clamp
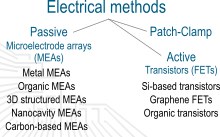
The most established electrical method for the investigation of electrogenic cells is the patch clamp technique. In this technique, a small needle penetrates the cell membrane and measures the intrinsic potential drop. While this method enables very high signal amplitudes it is not suitable for long-term measurements, with its invasive nature making it single-use only.
MEAs
Microelectrode arrays are the gold standard in the field of extracellular recordings and stimulation. A large variety of MEAs can be fabricated, using standard gold, more advanced porous materials, or using graphene. We improve the charge transfer of MEAs by testing new materials and shapes, and characterize the stimulation range of different electrode designs. We also design MEAs for highly specific applications such as enabling bidirectional communication with retinal tissue.
FETs
Transistors are active devices used in modern electronics and can also be used for biological applications. They provide a high degree of freedom to fine-tune different parameters towards engineering a well-suited device. Such devices, fabricated using silicon, graphene, or organic materials, can record the neuronal signal with the advantage that they can be scaled down to the nanoscale and provide an intrinsic amplification. Therefore, this approach poses the ability of creating hyper-high density device arrays which, in a long-term goal, can be implantable into the brain tissue. We are working with different concepts, ranging from field effect transistors to organic electrochemical transistors to establish the ideal architecture. Transistors are promising to provide the answers for human society on how our brain actually works and maybe even partially substitute some brain activity, i.e. for the treatment of Parkinson’s disease.
Measurement set-up
To be able to record signals from the developed arrays, there is the need to develop a measurement set-up which is able to measure all the electrodes simultaneously with a good time resolution below milliseconds. Our pico-amp set-up was developed for this purpose. The signal is recorded simultaneously on up to 64 channels. All devices can be characterized directly in the set-up in order to find optimal measurement conditions. The recorded signal can be amplified with a variable amplification factor and then be converted to a digital signal in order to evaluate the recorded data. With this set-up, it is possible to analyze cell systems like neurons and cardiomyocytes over long periods of time as well as perform repeated measurements to correlate different biological conditions as well as different electrode architectures.
Flexible and implantable electronics
With the ultimate goal to treat neurodegenerative diseases, to restore lost sensorimotor functions, and to understand the complex neural networks within the central and peripheral nervous system, we are developing neural interfaces capable of monitoring and modulating neural activity. Up to date, one of the main challenges in neural technology is the immune response that leads to implant rejection when using stiff implants, such as silicon-based probes. The latter, affects in turn the stability of the devices when implanted for long periods of time (months) in in-vivo applications. In our institute, we are developing flexible and compliant neural implants for a seamless integration with the body, thereby reducing the mechanical mismatch between prosthetic neural devices and our target nervous tissues.
To this end, we try to tackle the problem from different angles. First, we customize designs that match the implant with the target application. Furthermore, we fabricate neural implants based on polymeric materials and MEMS-based technology, procuring also different electrode coatings to achieve proper electrochemical capabilities. Additionally, implants are characterized to fulfill mechanical, electrochemical, and biological requirements, allowing us in turn to implement different neural interfaces from design, fabrication, and characterization to its final implementation. Thus, we are currently working on the development of different neural applications, from brain and peripheral nerve implants to miniaturized visual prosthesis.
High-density electronics
Another important aspect when recording neural activity in living tissue is the density of recording sites on the microelectronic array. With larger inter-contact distances, it is difficult to isolate the activity of individual neurons because each contact can be affected by the activity of multiple cells around the array. High-density arrays can resolve this issue by providing a detailed picture of the local activity patterns which enables us to isolate the activity of many individual neurons. For our neural recordings in intact brains, we therefore use high-density electrode arrays to study the function of large neural networks across different brain areas. This provides us with a unique view on local neural networks dynamics and the long-range communication across different brain areas. In our neural recordings in mice, we focus on understanding on how neural networks in the brain process sensory information to enable the generation of a corresponding behavioral response.
Dr. Simon Musall
Tel.: +49-2461-61-3539
e-mail: s.musall@fz-juelich.de
Prof. Dr. Andreas Offenhäusser
Tel.: +49-2461-61-2330
e-mail: a.offenhaeusser@fz-juelich.de
PUBLICATIONS:
Single-Trial Neural Dynamics Are Dominated by Richly Varied Movements, S. Musall et al., Nature Neuroscience 22, 1677-1686, October 2019
Toward a Bidirectional Communication Between Retinal Cells and a Prosthetic Device–A Proof of Concept, Rincón Montes et al., Frontiers in neuroscience 13, April 2019
Fabrication of ultrathin and flexible graphene-based devices for in vivo neuroprosthetics, Kireev et al., MRS advances 3, 1621-1627, June 2018
Graphene field effect transistors for in vitro and ex vivo recordings
D. Kireev et al., IEEE Trans. Nanotechnology 16, 1 (2017)
Graphene Multielectrode Arrays as a Versatile Tool for Extracellular Measurements
Kireev et al., Advanced Healthcare Materials 6 (2017)
An evaluation of extracellular MEA versus optogenetic stimulation of cortical neurons
Maybeck et al., Biomed. Phys. Eng. Express 2, 5 (2016)
Versatile Flexible Graphene Multielectrode Arrays
Kireev et al., Biosensors 7, 1 (2016)
Nanostructured cavity devices for extracellular stimulation of HL-1 cells
Czeschik et al., Nanoscale, 7, 9275 (2015)
Sensitivity enhancement of Si nanowire field effect transistor biosensors using single trap phenomena
Li et al., Nano Letters 14, (2014)
On Chip Guidance and Recording of Cardiomyocytes with 3D Mushroom-Shaped Electrodes
Santoro et al., Nano Lett. 13, 5379 (2013)
Nanostructured gold microelectrodes for extracellular recording from electrogenic cells
Brüggemann et al., Nanotechnology 22, 26 (2011)
Nanocavity electrode array for recording from electrogenic cells
Hofmann et al., Lab Chip 11, 1054 (2011).
Graphene Transistor Arrays for Recording Action Potentials from Electrogenic Cells
Hess et al., Advanced materials 23, 5045 - 5049 (2011)