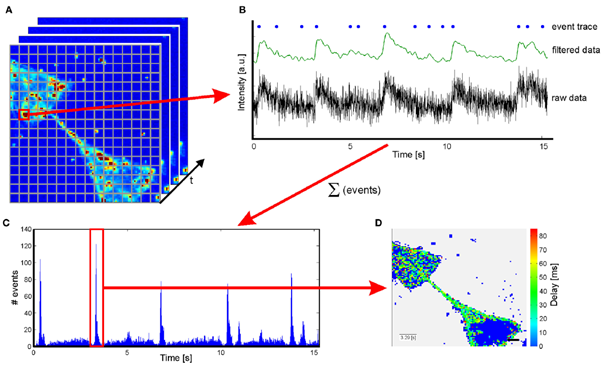
Calcium Imaging
The firing of an action potential is accompanied by an influx of calcium ions into the cell. This increasing intracellular calcium ion concentration can be detected by fluorescent dyes (e.g. Fluo-4) or genetically encoded fluorescent sensor proteins (e.g. GCaMP), that have been introduced into the cell beforehand, allowing video recording of neural activity.
One way is to load the cell culture with a dye like Fluo-4-AM (Invitrogen). Upon cellular activity the Ca binds to Fluo-4 leading to a conformational change and the increase in fluorescence. The other method is to introduce a genetically encoded calcium sensor like GCaMP into the cell. GCaMP consists of calcium binding domains and a circularly permuted GFP. Similar to the Fluo-4 the protein changes its conformation upon Ca2+ binding leading to an increase in fluorescence. While using Fluo-4 is a fast and comparable easy method, GCaMP requires a well-planned experimental setup. However using a genetically encoded sensor provides more opportunities in selectively image special neuronal types.
To process the calcium imaging movies we established an analysis software written in MATLAB for automatic ROI detection and signal propagation. With this software we are able to extract the important data like propagation direction and network synchrony.
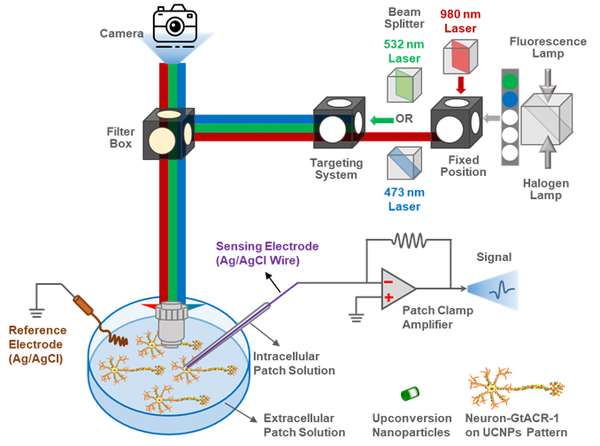
Upconverting Nanoparticles (UCNPs)
Patterns of UCNPs control neuronal growth in vitro. Simultaneously, they can convert infrared light into the visible spectrum necessary for opening optogenetic channels. Our custom built system allows targeting various cells with 473nm or 532nm laser lines, as well as providing a fixed laser point of 980nm all concurrent with wide-field fluorescent imaging and electrophysiological measurements.
CONTACT:
Prof. Dr. Andreas Offenhäusser
Tel.: +49-2461-61-2330
e-mail: a.offenhaeusser@fz-juelich.de
Dr. Vanessa Maybeck
Tel.: +49-2461-61-3285
e-mail: v.maybeck@fz-juelich.de